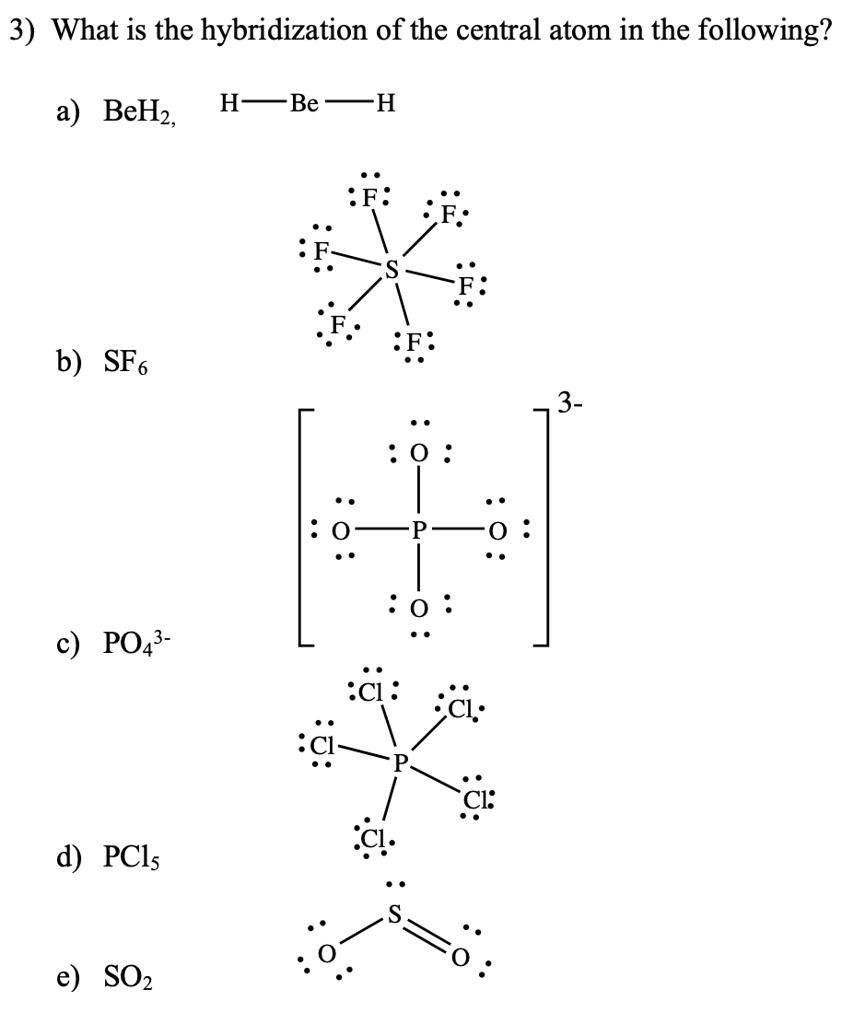
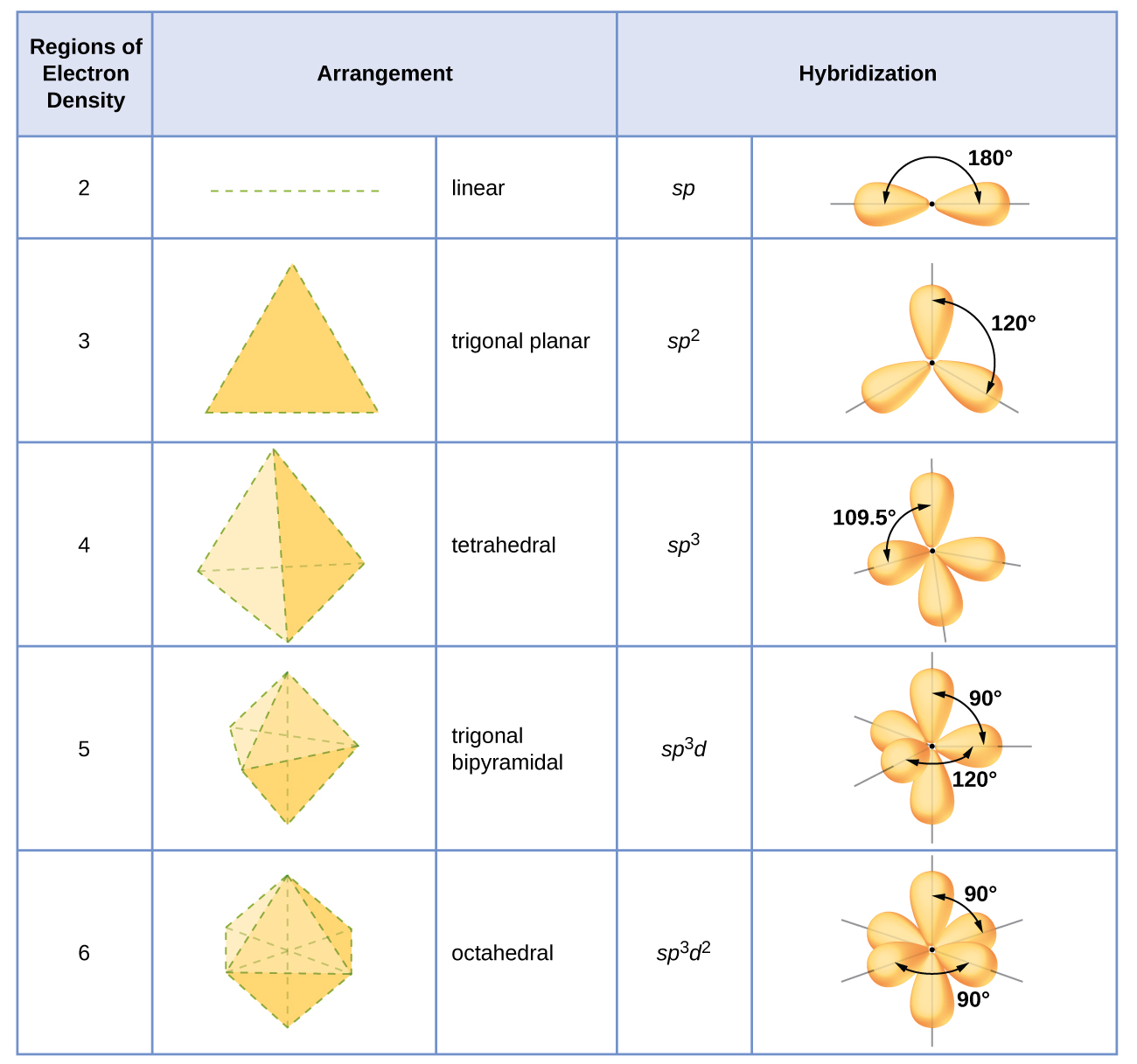
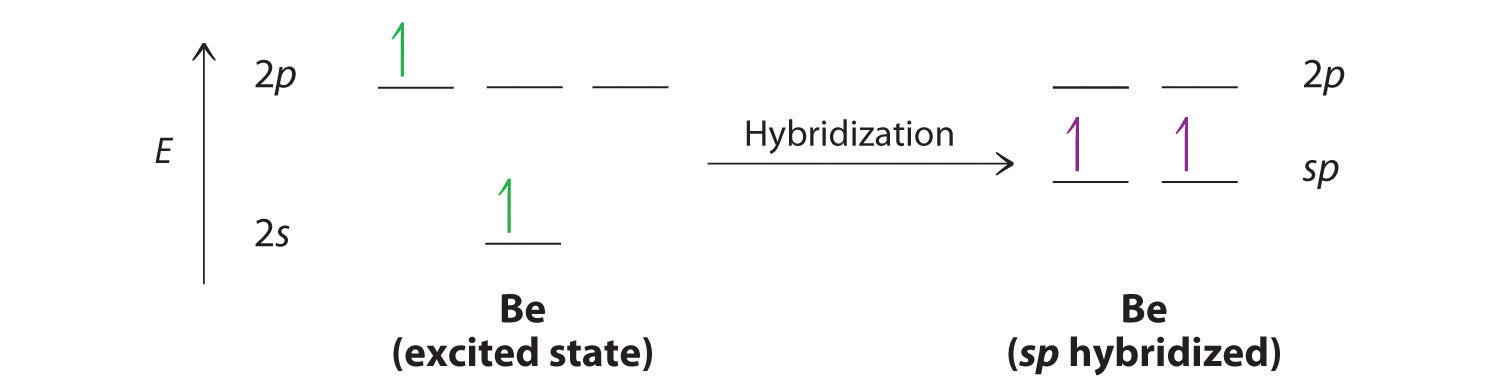
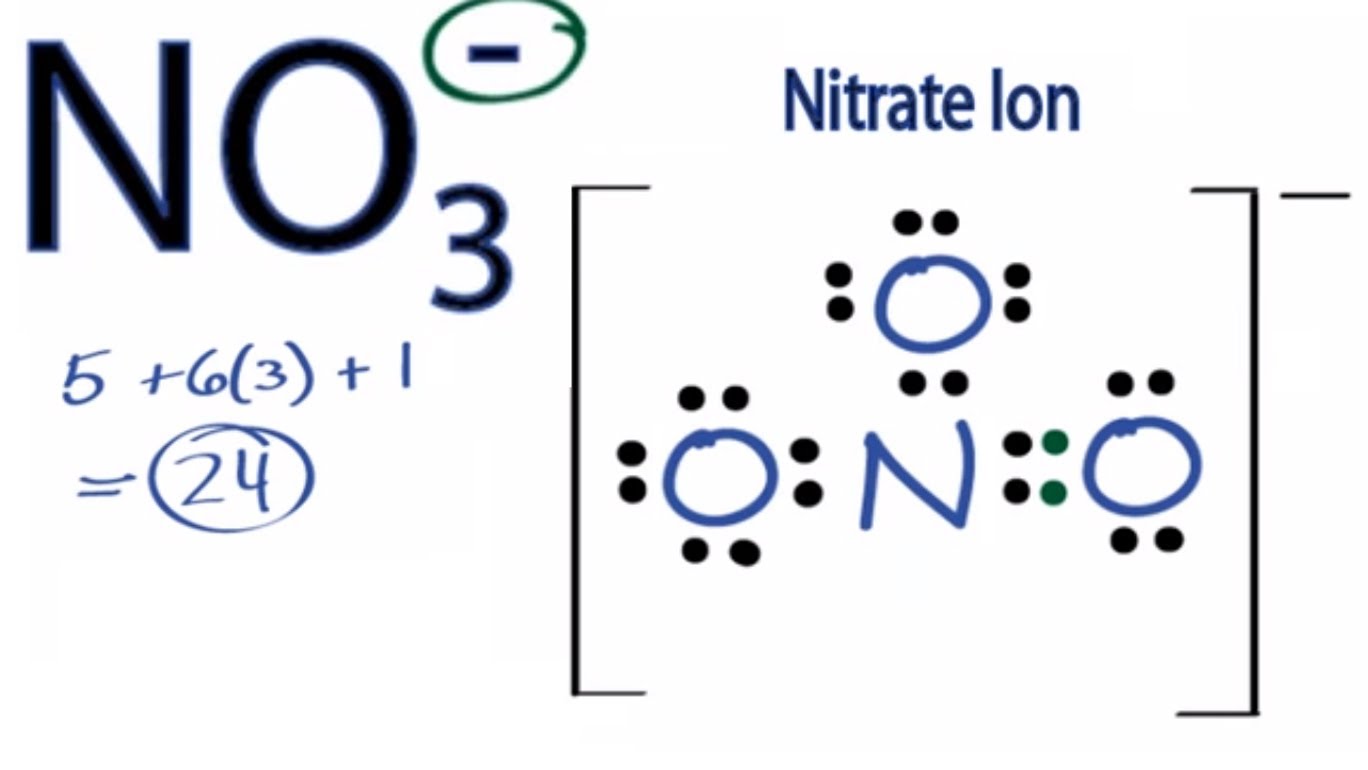
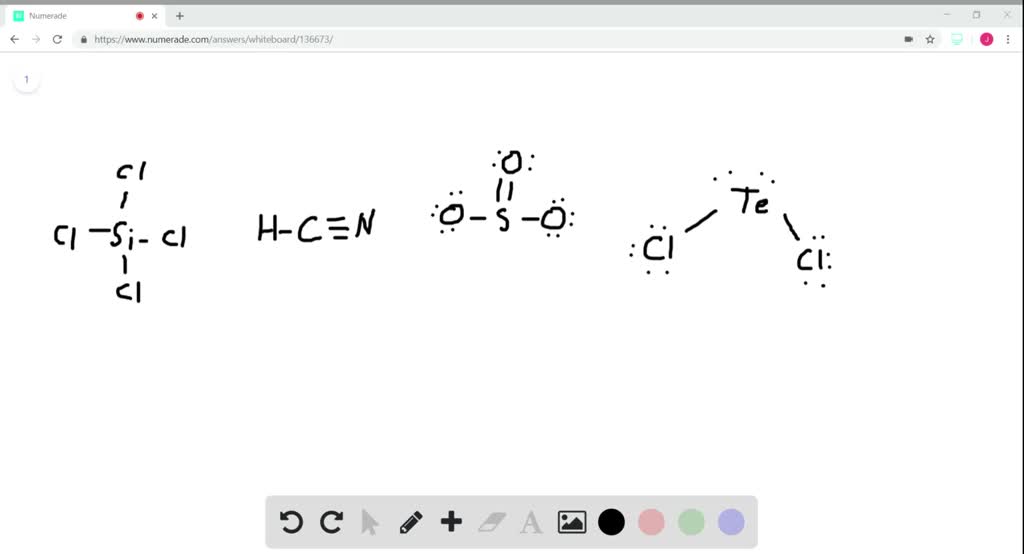
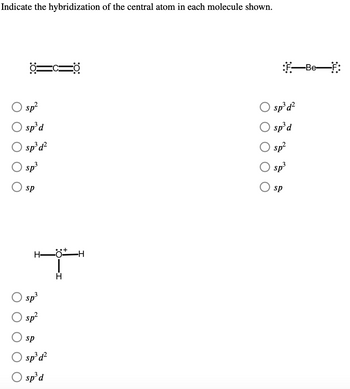
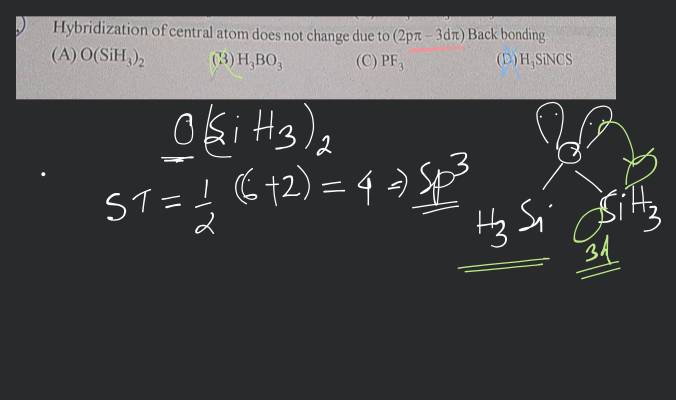19. In which one of the following species the central atom has the type of hybridization which is not the same as that present in other three? 1)SF4 2)I3 3)SbCl3 4)PCl5

Specify the hybridization of the central atom in the following species respectively N 3 , NOCl , N 2 O :A. sp , sp 2, spB. sp 2, sp , spC. sp 2, sp 2, spD. sp , sp , sp 3

What hybridization is generally utilized by the central atom in a square planar molecule? - CBSE Tuts
Hybridization of central atoms in the moleculesN(CH3)3 and N(SIH3) respectively are(1) sp2 and sp2(2) sp3 and sp3(3) sp2 and sp34) sp3 and sp2