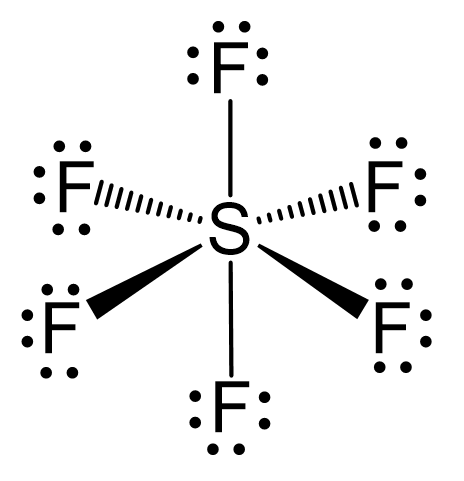
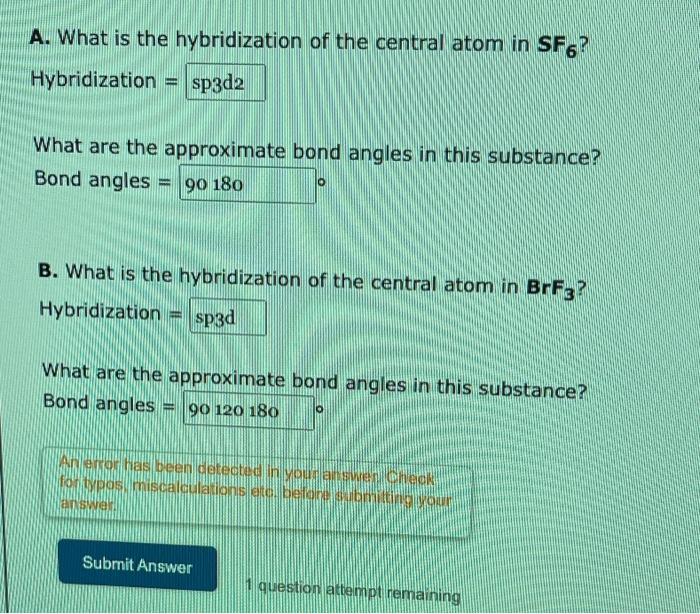
Chemistry - Molecular Structure (35 of 45) s-p3-d2 Hybridization - Sulfur Hexafloride - SF6 - YouTube

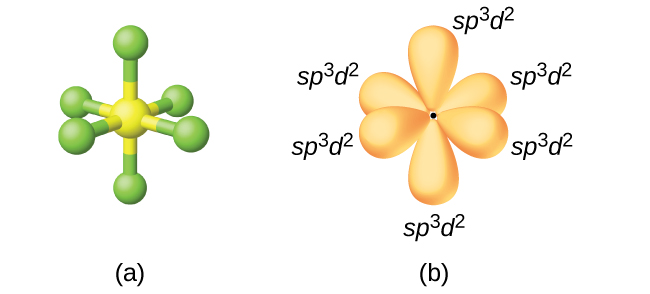
The sp3d2 Hybridization and Octahedral Geometry | Molecular geometry, Chemistry, Electron configuration

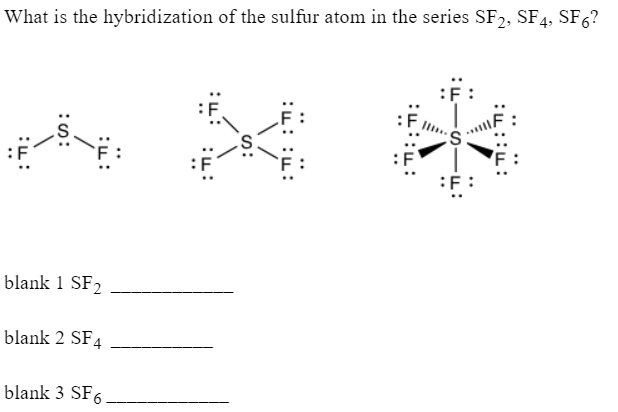
What is the hybridisation of SF6 whats the method to find it - Chemistry - Chemical Bonding and Molecular Structure - 5272961 | Meritnation.com

Chemistry - Molecular Structure (35 of 45) s-p3-d2 Hybridization - Sulfur Hexafloride - SF6 - YouTube

What atomic or hybrid orbitals make up the sigma bond between S and F in sulfur hexafluoride, SF6? | Homework.Study.com

Sp3d2 Hybridization | Formation Of SF6 Molecule | Chemical Bonding,Bsc.1st Year,Inorganic Chemistry - YouTube

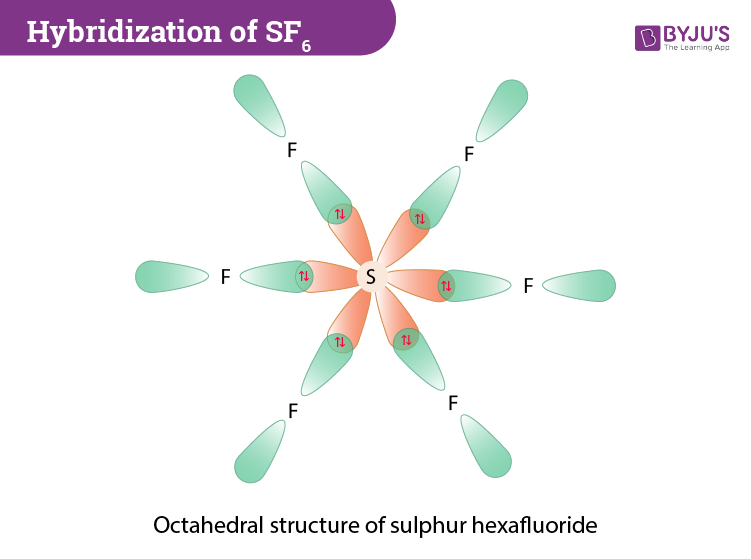
![Solved] In SF6 the hybridisation of sulphur is: Solved] In SF6 the hybridisation of sulphur is:](https://storage.googleapis.com/tb-img/production/21/01/F1_Utkarsha_15.1.21_Pallavi_D27.png)