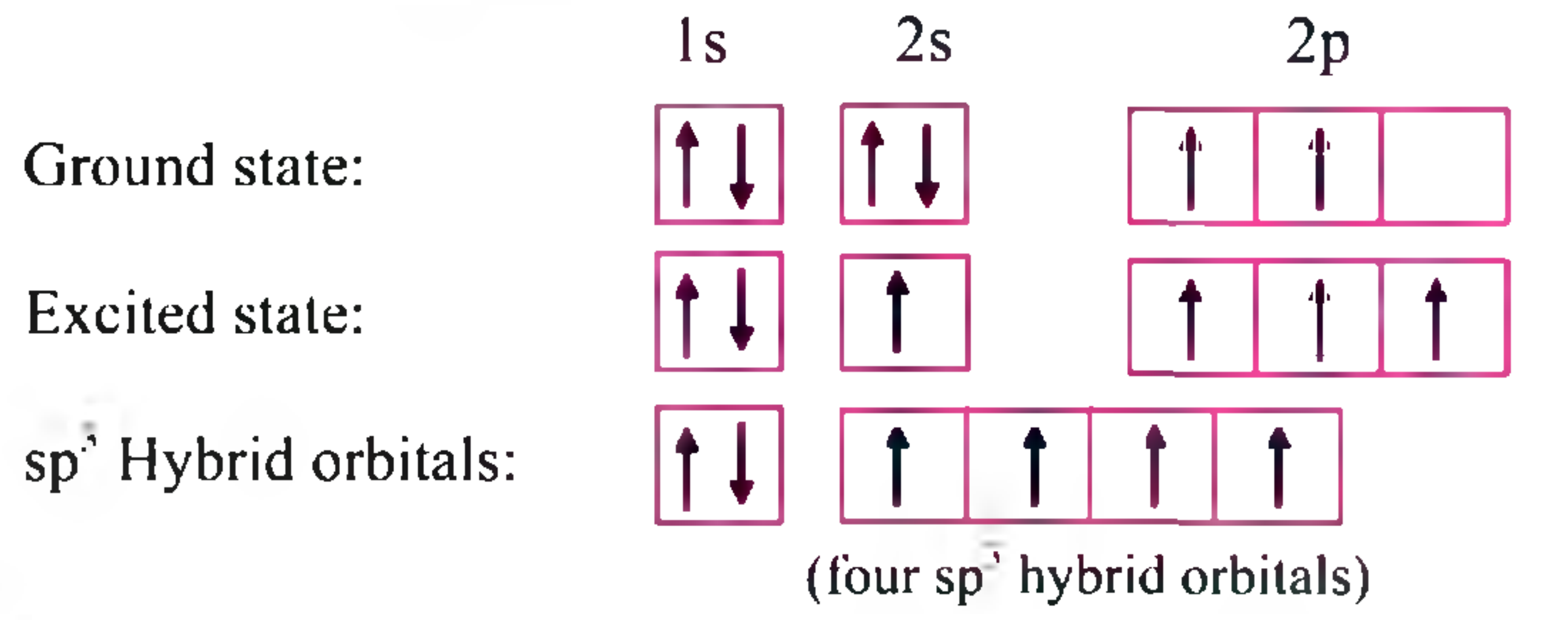
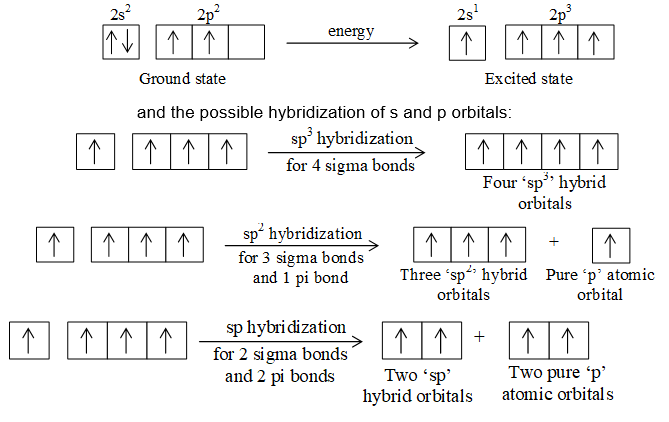
What is the hybridization of carbon 1 and carbon 3 in the molecule on the right? How many total sigma and - brainly.com
D response graph showing the effect of pH buffer, NaCl concentration,... | Download Scientific Diagram
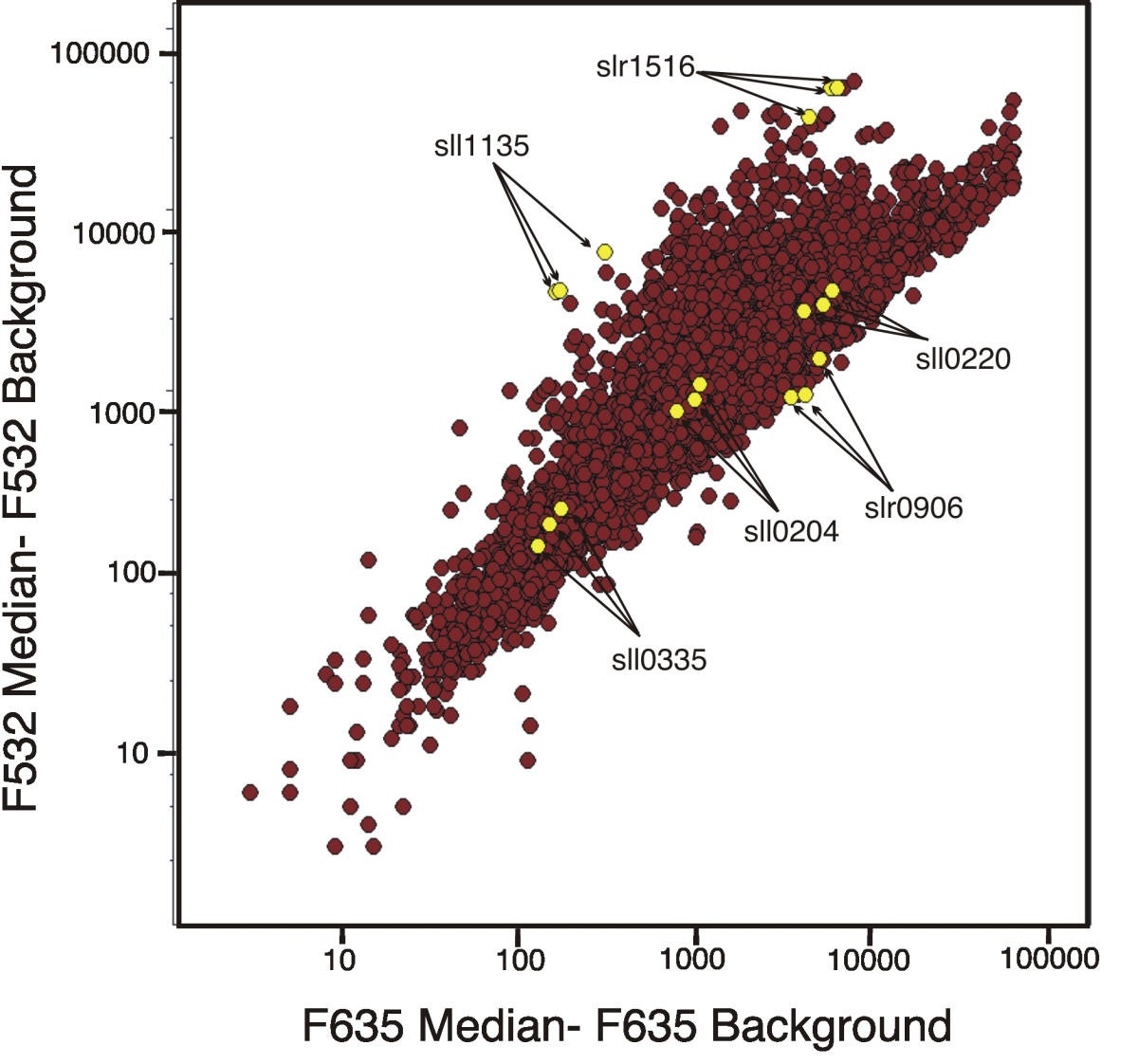
The construction and use of bacterial DNA microarrays based on an optimized two-stage PCR strategy | BMC Genomics | Full Text

The effects of NaCl concentrations in hybridization buffers on final... | Download Scientific Diagram

The Role of Repulsion in Colloidal Crystal Engineering with DNA | Journal of the American Chemical Society

Exotic Two-Dimensional Structure: The First Case of Hexagonal NaCl | The Journal of Physical Chemistry Letters








![BH001] Hybridization Solution I | Biosolution BH001] Hybridization Solution I | Biosolution](http://biosolution.cafe24.com/wp-content/uploads/2015/05/BH001-Hybridization-Solution.jpg)